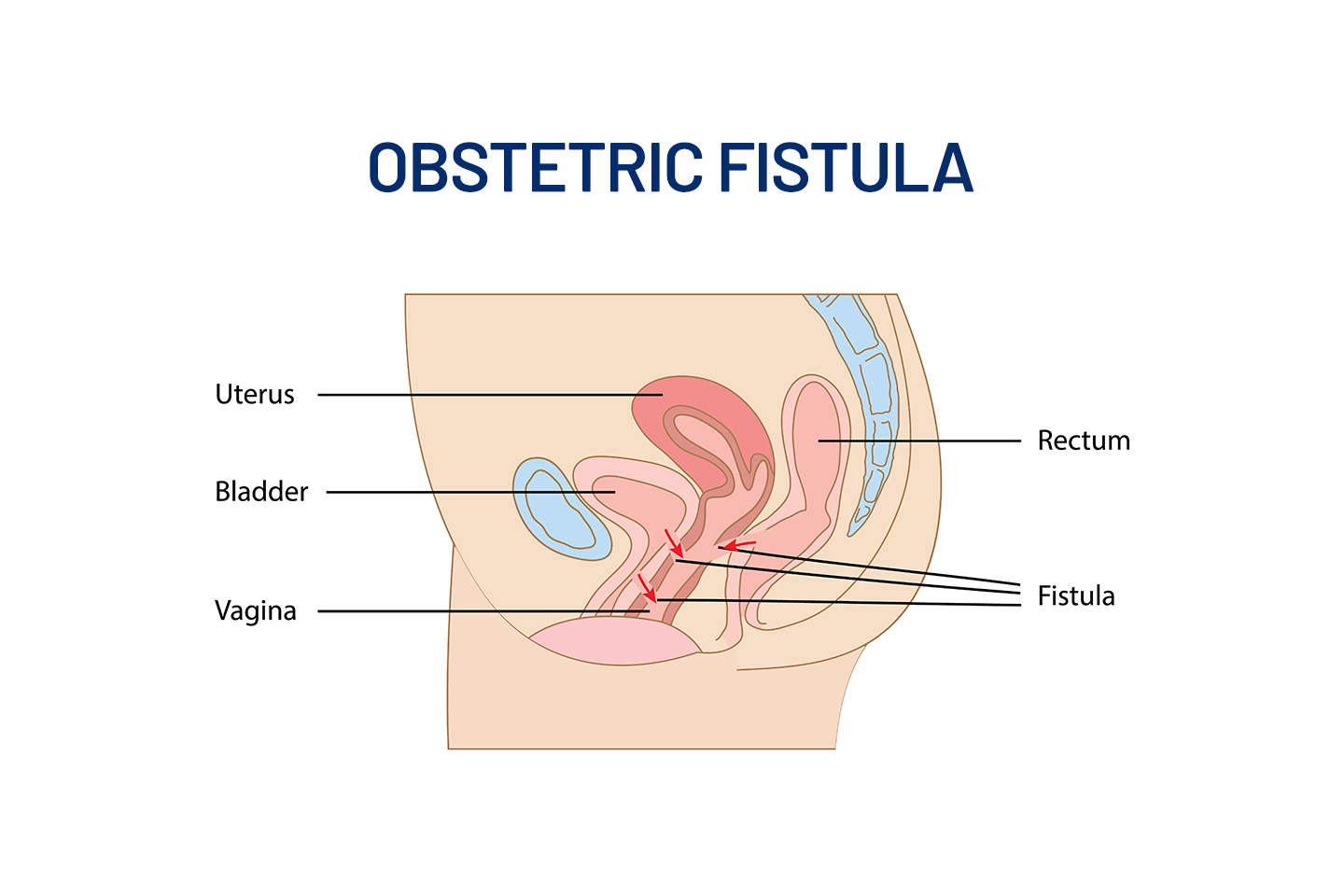
Medical Devices
Handheld Robotic surgery v/s traditional laparoscopic surgery
April 20, 2024
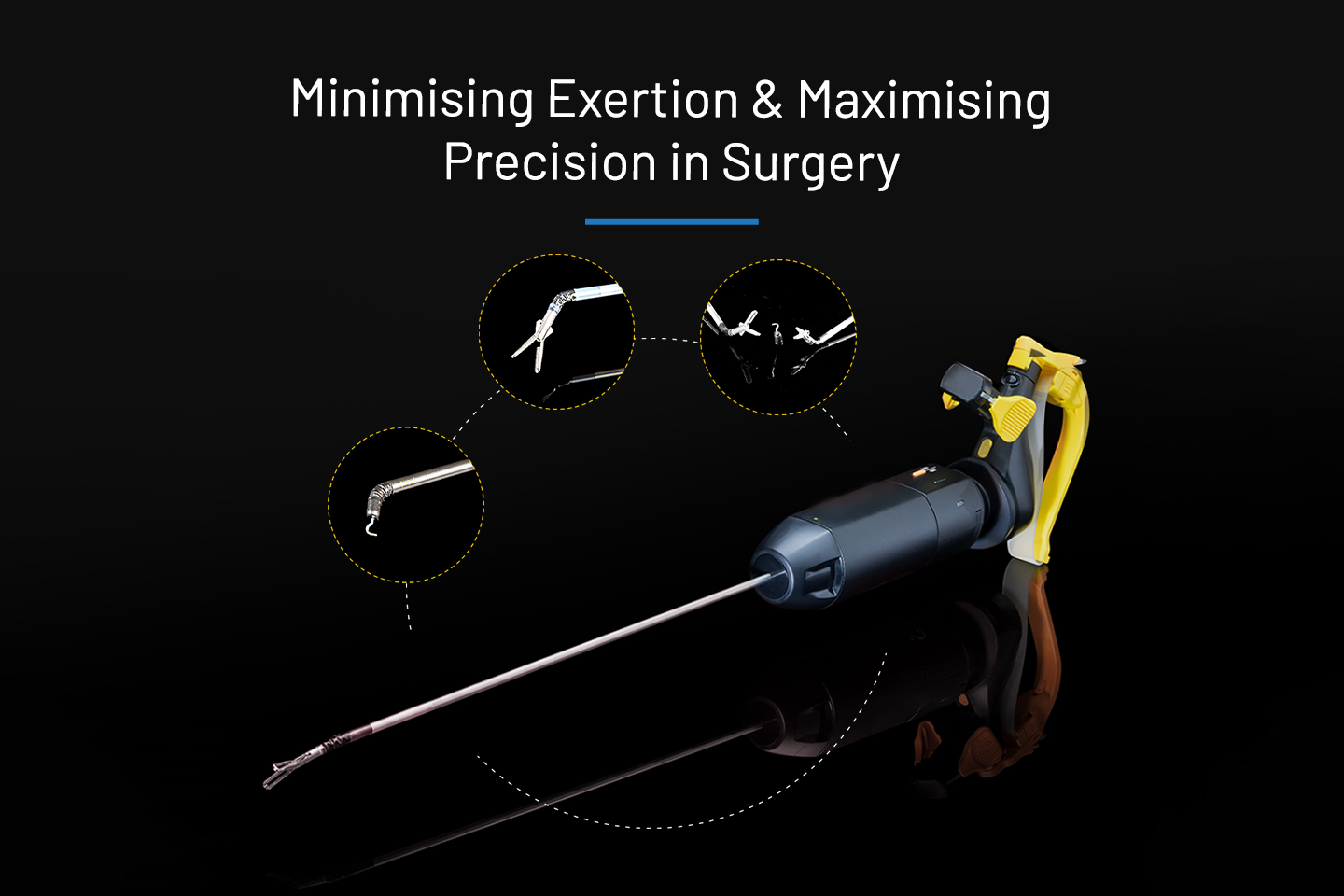
Explore Handheld Robotic Surgery vs. Traditional Laparoscopic Approach: Discover the advancements, benefits, and differences between handheld surgical devices and traditional laparoscopic methods.
Medical Devices
Active Robotics in Knee Replacement Surgeries for varying patient anatomies
April 12, 2024
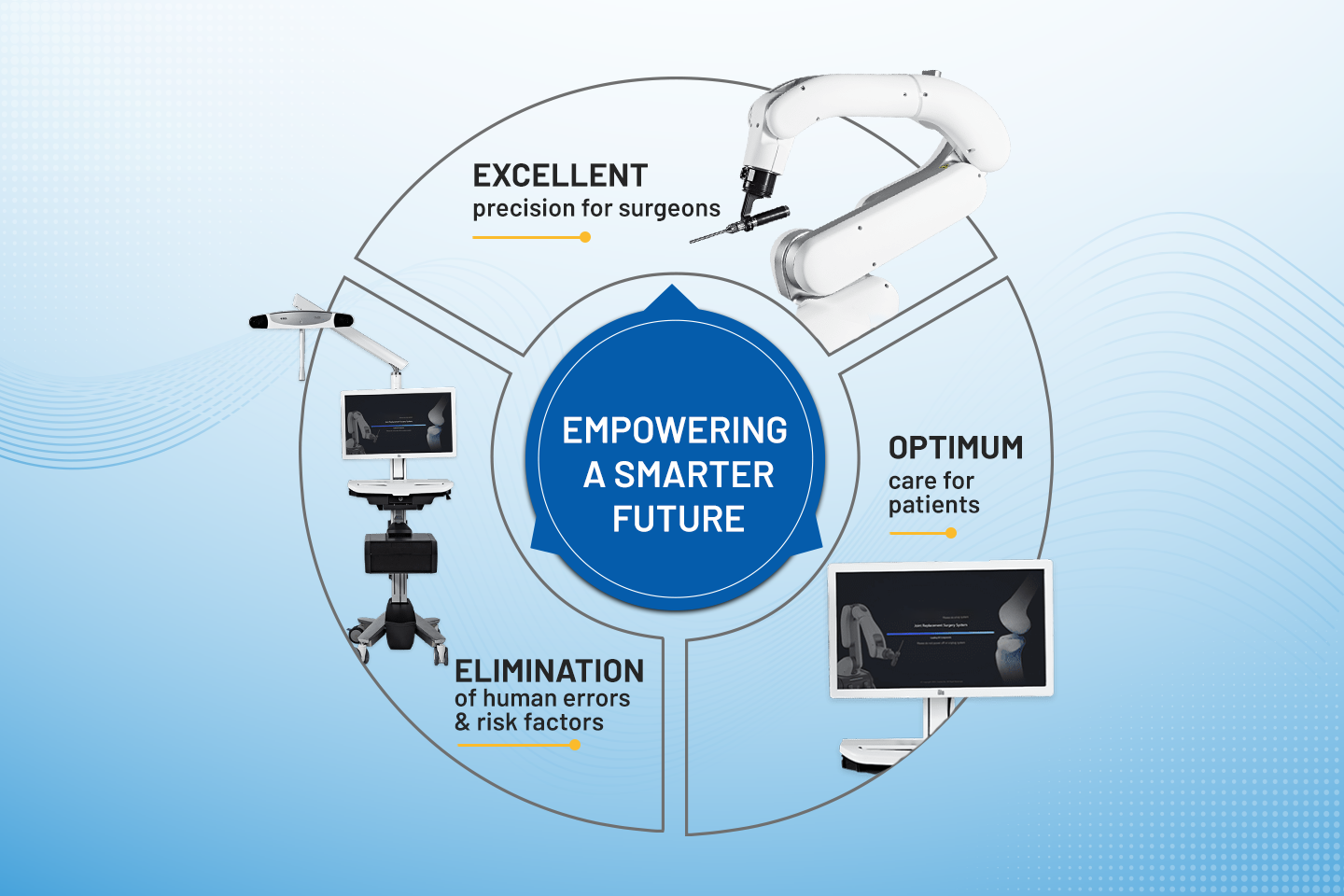
Explore how active robotics is revolutionizing knee replacement surgeries, catering to diverse patient anatomies for enhanced precision and outcomes.
Medical Devices
Dengue Resurgence: What Patients Should Know About Dengue Testing and Test Kits
April 06, 2024

Stay informed during the Dengue resurgence. Explore our guide on Dengue testing and test kits, empowering patients with valuable insights for informed decisions. Learn about the essentials to navigate through this challenging time.