
HARNESSING THE POWER OF HYBRID
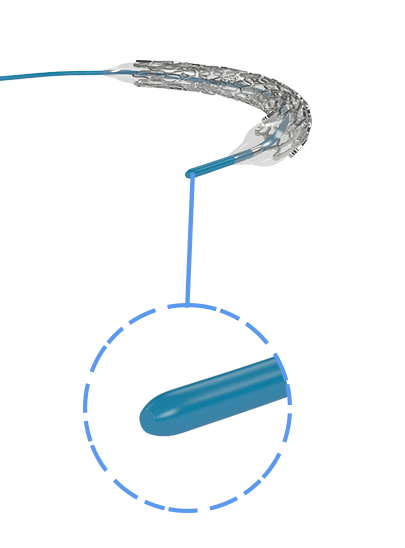
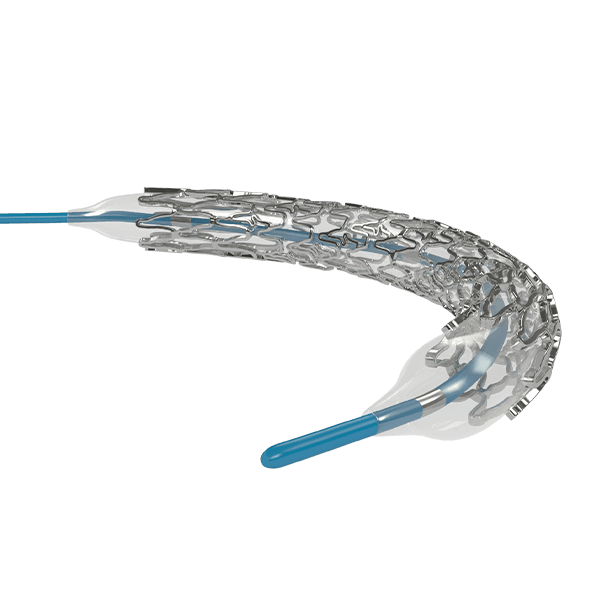
Myra™ BMS comprises of the following components:
- A balloon-expandable L605 Cobalt Chromium Peripheral Stent
- An over-the-wire stent delivery PTA Balloon Dilatation Catheter
The stent is pre-mounted on Balloon Catheter & placed between two platinum-iridium radiopaque markers bands.
Key Features
Highlights
- Hybrid stent architecture offers strength & flexibility
- Short balloon overhang facilitates tracking and crossing of the stent delivery system
- Longitudinal flexibility to cross tortuous vessels and aortic bifurcation with contra-lateral approach
- Minimal foreshortening (<1.0%) ensures accuracy of placement
- 1.5–9 mm sheath is compatible with 6 Fr, and the 10 mm sheath is compatible with 7 Fr.
Indication
Intended for use in the treatment of atherosclerotic diseases of peripheral arteries below the aortic arch, with reference vessel diameter of 5.00 mm to 10.00 mm for Iliac / Sub-Clavian and for other protected peripheral arteries in patients eligible for Percutaneous Transluminal Angioplasty (PTA) and Stenting procedures.
FAQs
1. What is Myra™ BMS?
2. Where can Myra™ BMS be used?
3. What is special about its material?
4. Does Myra BMS provide accurate placement?
5. How is visibility ensured during the procedure?