Introduction
The replacement procedure- also known as MVR replacement - is generally carried out when the patient suffers from mitral valve stenosis or mitral valve regurgitation.
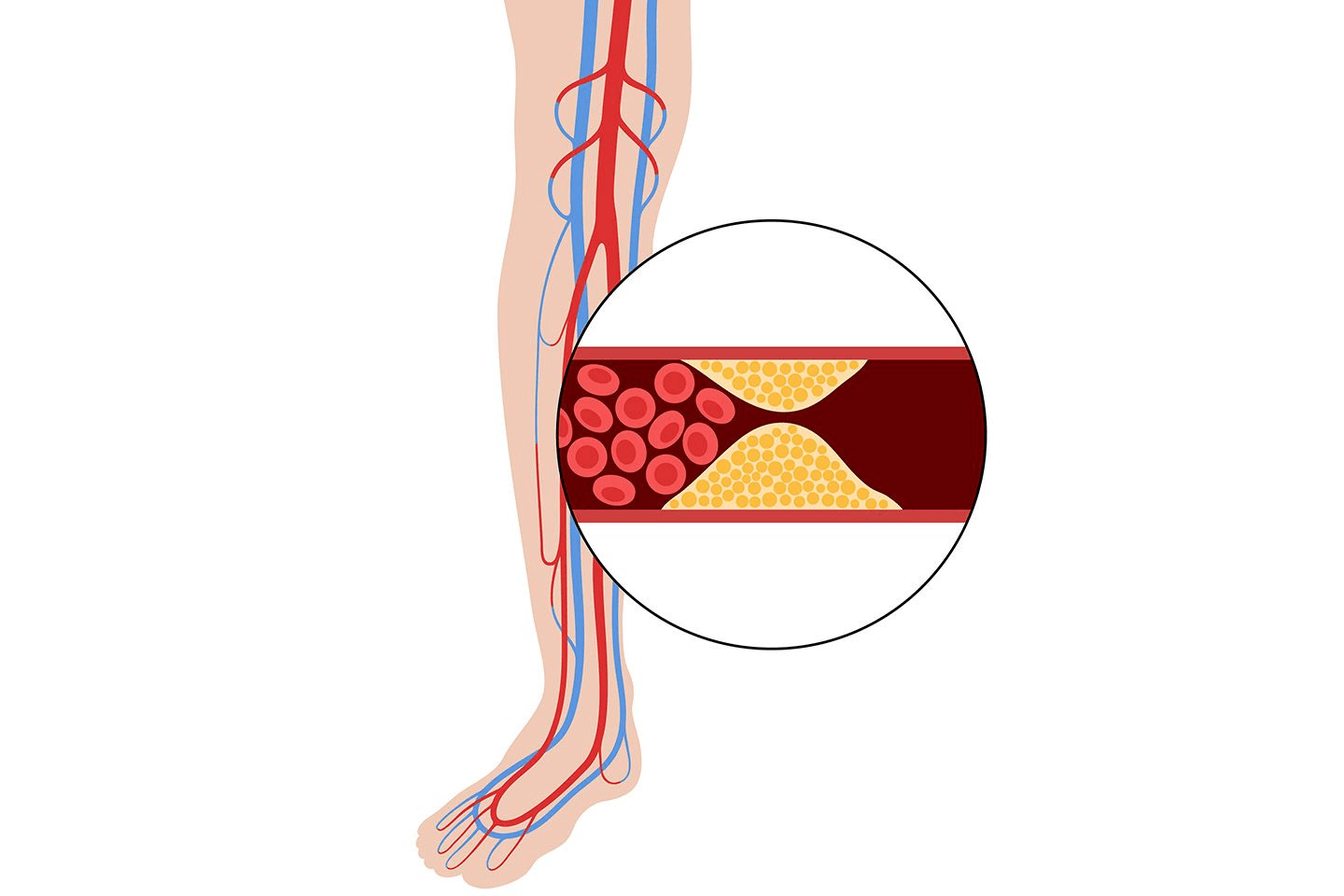
Stenosis- The opening of the mitral valve is narrowed and as a result the valve does not open properly. This restricts the flow of blood from the left atrium to the left ventricle.
Regurgitation- The mitral valve does not close tightly and the blood flows backward in the heart.
What happens during a mitral valve replacement surgery?
The patient must stop consuming food and liquids for a few hours prior to the mitral valve replacement procedure. They must also stop consuming certain medications, like blood thinners.
MVR replacement is carried out under general anesthesia. The cardiac surgeon puts the patient on a special machine called a heart-lung machine. The surgeon makes a large incision on the chest and removes the mitral valve before replacing it with the artificial valve. When the procedure has been completed, the heart-lung machine is taken off and the patient gradually wakes up from the anesthesia-induced sleep.
This procedure can also be performed through robotic surgery.
A mitral valve replacement is a major surgery, and the patient has to stay in hospital for a few days while recovering. Upon discharge, the patient needs to be assisted home. Once home, one is advised to follow the doctor's instructions for the post-operative care and healing of the wound. It is important to continue to rest and not undergo physical exertion for at least a few weeks.
After discharge, one has to visit the doctor for the removal of the stitches and thereafter undergo follow up visits at regular intervals as advised and instructed by the doctor.
Are there any complications associated with MVR replacement?
A mitral valve replacement may have certain complications, such as infection and clotting. There have also been reports of valve failure. The doctor will discuss all possible outcomes and asks to come in for frequent check-ups to evaluate the condition of the valve.
Myval THV from Meril
The Myval transcatheter heart valve has been indigenously developed by Meril Life Sciences Pvt. Ltd. With low device related mortality and low risk of stroke, Myval THV offers a lot of precision in intended outcomes. Noteworthy features of this device include internal PET sealing cuff for minimizing PVL and puncture resistance and cobalt alloy frame for radiopacity and high radial strength.
The Myval THV has a hybrid cell design with open cells for un-jailing of coronary ostia and closed cells that offer high radial strength.
Meril continues to pioneer research in the field of medical technology to alleviate human suffering. Founded in 2006, the company is present in 100+ countries with a staff of 4000+ employees.
Conclusion
Advances in medical science have now made it possible to successfully treat and manage conditions affecting the mitral valve disease. For the best outcomes, follow your doctor's advice on post-operative care for the early recovery and also maintaining healthy heart. If you smoke, make sure to quit. Follow a diet that is healthy for the heart. Your doctor will guide you with a diet plan to help you stay in shape.
SHARE NOW