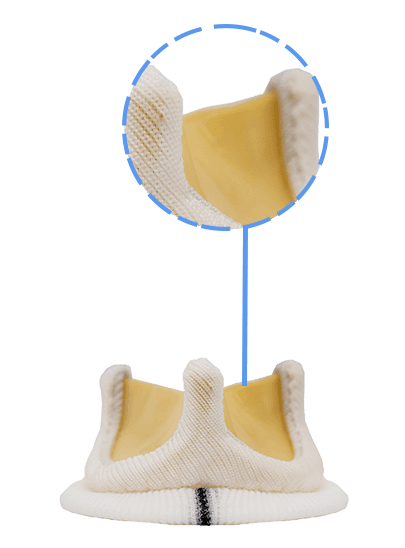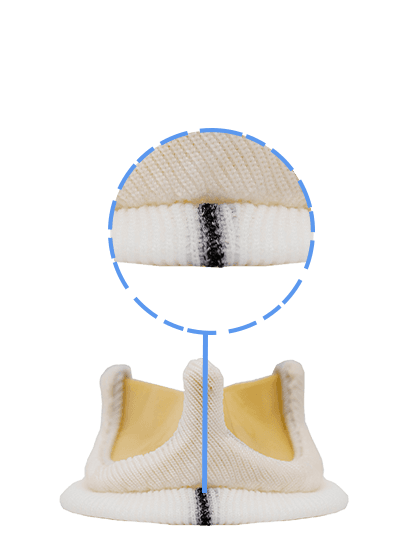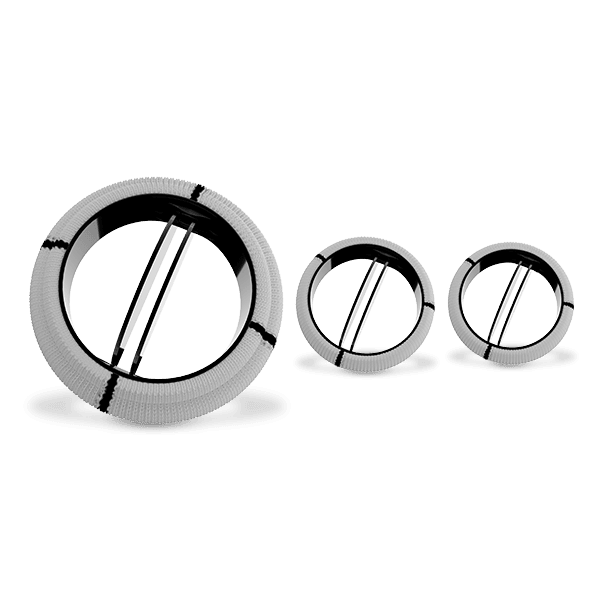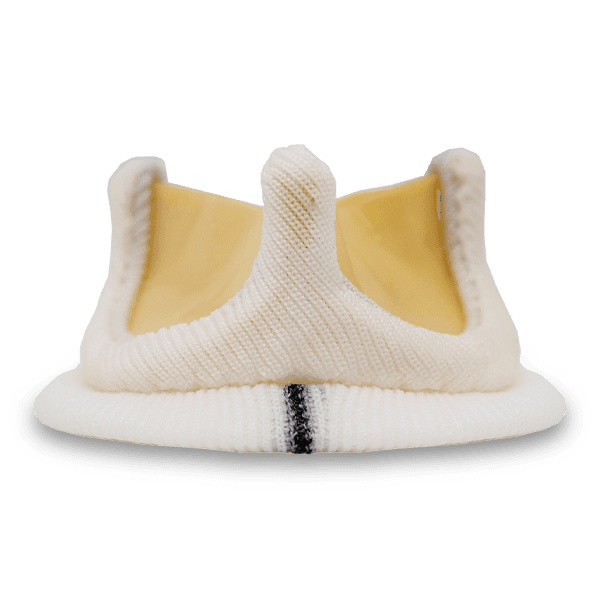
Dafodil Neo™ Pericardial Bio prosthesis is a Triple Composite biomechanically engineered valve. It combines the latest engineering advancements over the Dafodil valve along with the benefits of bovine pericardial tissue. Dafodil Neo™ comes with certain enhancements in implantability (reduced profile and an extension holder handle) suitable for minimally invasive valve replacement.
A reduction in the sewing ring diameter ensures better utilization of the patient annulus area that ultimately translates into better hemodynamics for the patient. Dafodil Neo™ Aortic valve comes with a unique 17 mm size offering (by prescription only) that is suited for narrow aortic annulus found in certain patients.
Pericardial tissue and Elgiloy stent material are both known for their optimal intrinsic properties for valve manufacture. Integrated processes such as Unique Tissue fixation, Advanced Tissue Thickness Matching Technology for Uniformity, and Tissue Deflection Testing enable leaflets to be accurately matched.
Position-specific design answers the challenging requirements of the mitral & aortic position Unique Delivery System For Mitral Position with the new Active flex technology that helps in reducing the external stent post diameter to aid in valve placement in the mitral position.
Key Features
Trusted durability Properties
Ease of Implantability Properties
Hemodynamics Properties
Indication
An Aortic Dafodil Neo™ Pericardial Bioprosthesis is indicated for use in patients whose aortic valvular disease is sufficiently advanced to warrant replacement of their natural valve with a prosthetic one. It is also intended for use in patients with a previously implanted aortic valve prosthesis which is no longer functioning adequately and requires replacement. In the latter case, the previously implanted prosthesis is surgically excised and replaced by the replacement prosthesis.