Transcatheter Aortic Valve Replacement (TAVR)
Understanding the problem in the heart valve
The human heart is comprised of 4 chambers as well as 4 valves. These valves make certain that the flow of blood through the heart in the right direction.
In the following conditions, the proper functioning of the valves get hampered:
- In case the heart valve gets narrowed to a large extent or gets incapacitated, not allowing sufficient blood flow through it, it is called as valve stenosis.
- Leaking valves are called as “regurgitant valves”.
- Numerous other valve problems occuring as a result of age-dependent causes or infective diseases such as rheumatic fever and infective endocarditis.
What is known as Valve Stenosis?
In case a heart valve is opening inadequately because of thickened leaflets, stiffness or fusion of its leaflets, it is known as “Stenosis” or a Stenosed Valve. As a result of this irregularity, the heart needs to work more for pumping adequate blood through the valve, or the body may undergo a decreased oxygen supply, demonstrating heart symptoms.
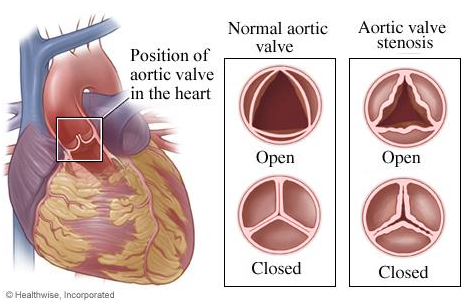
What is Aortic Valve Stenosis (AS)?
As the name suggests, aortic valve stenosis (AS) is amongst the most frequently reported and most severe form of valve disorders. The aortic valve is located on the heart’s left side and opens while the blood is being pumped out from the heart to the entire body. AS is a contraction of the opening of aortic valve, limiting the blood flow from the lower left chamber of the heart (left ventricle) to the main artery that carries blood away from the heart to the rest of the body (aorta) and may also disturb the pressure in the heart’s upper left chamber (left atrium).
Though AS can occur as a result of a heart defect present at birth called a bicuspid aortic valve in some individuals, it occurs more frequently during advanced age, because of calcium build-up in blood vessels or accumulation of dead cells (scar formation), which harms the valve and decreases the blood flow through the valve.
Does aortic stenosis always produce symptoms?
AS does not show symptoms every time it occurs. A large proportion of AS patients remain without symptoms until the reduction of blood flow reaches worryingly low levels.
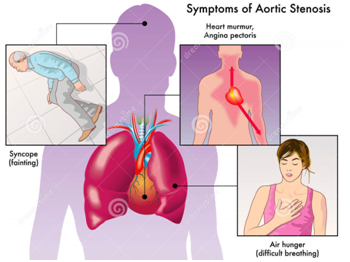
Symptoms of aortic stenosis may comprise of the following:
- Difficulty in breathing (dyspnea)
- Pain, pressure or tightness in the chest (angina).
- Temporary loss of consciousness caused as a result of sudden drop in blood pressure, referred as “syncope” or fainting.
- A feeling of heavy pounding in the chest or feeling of own heartbeats (palpitations)
- Decreasing levels of activity or decreased ability to perform normal activities necessitating only mild efforts (fatigue)
- Abnormal Heart Sounds during Stethoscope Examination
However, an AS patient may not always be symptomatic. However, if family members report reduced activity levels or notable tiredness, a doctor must be consulted and appropriate diagnostic tests must be undertaken.
Infants and children, suffering from valve defects from birth, may demonstrate symptoms such as:
- Tiredness upon effort
- Not gaining weight
- Not feeling hungry or not eating enough
- Difficulty in breathing
How does aortic stenosis progress or cause increasing problems?
Along with the AS symptoms, the wall of the heart’s lower left chamber may also demonstrate thickening of muscles due to additional effort to pump blood through the contracted valve opening.
The thickened wall accumulates more space within the lower heart chamber, allowing less scope for a reasonable blood supply to the body. Early treatment can aid in reversal or delay the disease progression.
Who is at risk for aortic stenosis?
Senior Citizens
Elderly individuals are mostly affected by aortic stenosis due to scar tissue formation and calcium buildup in the flaps of the aortic valve and also the vasculature. Age-associated AS generally starts after 60 years of age, but then again it frequently remains asymptomatic until 70 or 80 years.
Aortic stenosis in the young
A structural defect in the aortic valve, present at birth, is the most prevalent cause of aortic stenosis in young people. In such cases, usually only two cusps grow instead of the normal three, which is called a “bicuspid valve.”
Another important cause of AS in young individuals is the presence of a valve opening which does not grow along with the heart.
What treatments are advisable for people with aortic stenosis?
In case of absence of symptoms or presence of mild symptoms, a doctor may just recommend watchful waiting and regular follow-up in case of appearance of any symptoms. However, it is recommended that each aortic stenosis patient should be tested with a heart ultrasound for endorsing the most safe and best treatment option. On the contrary, irrespective of presence of symptoms, it may be sensible to go ahead with a suitable treatment on the basis of imaging test outcomes.
Likely treatments of aortic stenosis may include valve repair or valve replacement. If agreement is reached for proceeding with surgery (Surgical Aortic Valve Replacement or SAVR), it is vital to proceed with the procedure without any further delay.
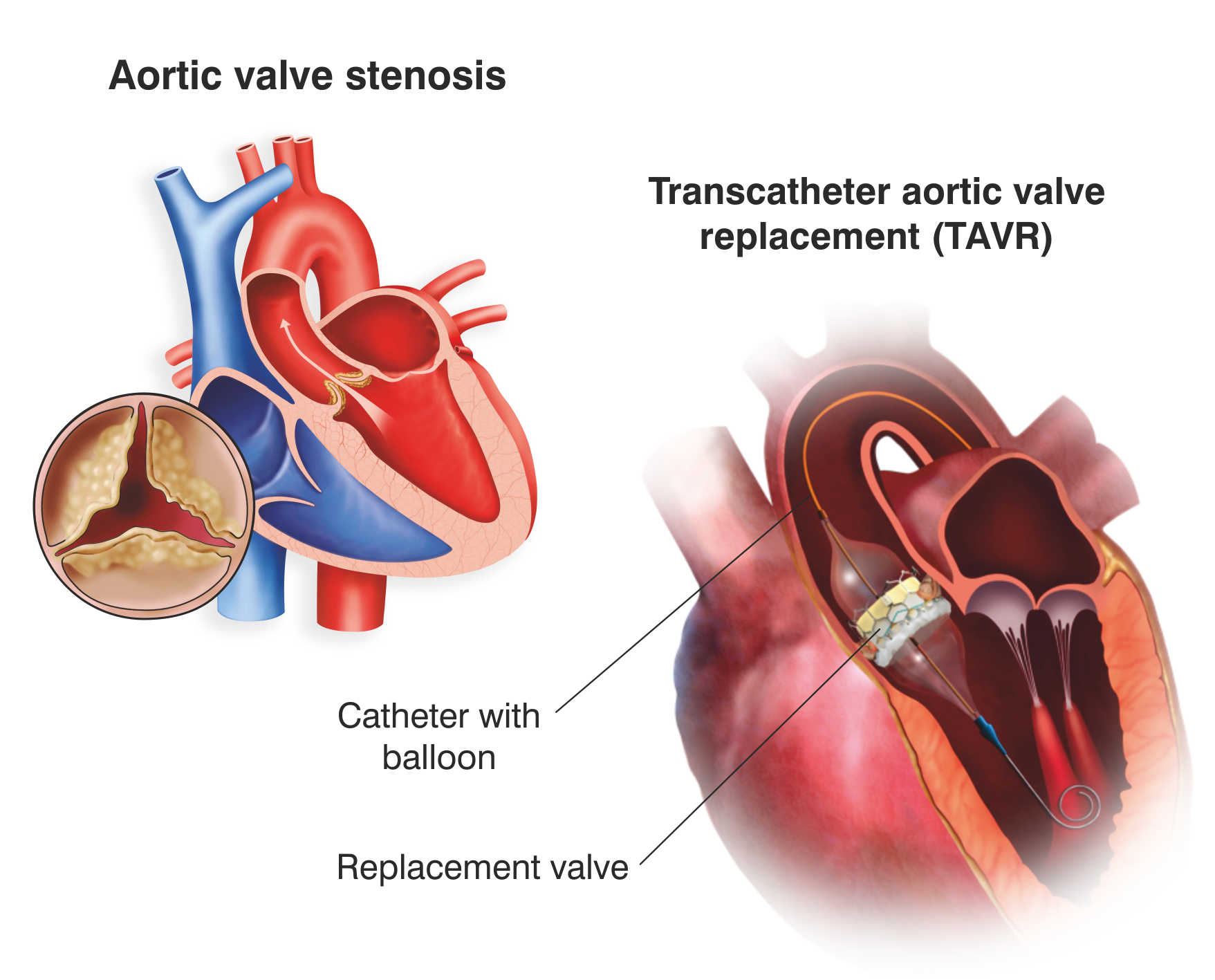
What is a Transcatheter Aortic Valve Implantation (TAVI) or Transcatheter Aortic Valve Replacement (TAVR)?
TAVI or TAVR is a marginally invasive procedure of surgery for replacing the inefficient/improperly functioning aortic valve with a brand new artificial valve.
Who is a good candidate for TAVR?
As per contemporary endorsements, TAVR procedure is reserved for patients who are deemed to be at high or intermediate risk for undergoing an open heart surgery (Surgical Aortic Valve Replacement/SAVR). On the basis of this reason, majority of the TAVR candidates belong to the age group of 70 to 80 years, often suffering from co-morbid clinical conditions. TAVR might also be an effective selection to improve quality-of-life in patients who else have restricted selections to repair their diseased aortic valve.
Risks with TAVR / TAVI
A TAVR procedure resembles a balloon angioplasty treatment or simply an angiogram in terms of time taken for performing the procedure and recovery period, and perhaps needs a comparatively brief duration of hospital stay versus open-heart surgery (approximately 3-5 days). Still, TAVR is not completely devoid of risks.
Some of the risks associated with TAVR are as follows:
- Bleeding
- Vascular complications
- Problems with the artificial valve
- Stroke
- Irregular heart beats/Arrhythmia
- Kidney disease
- Infection
- Heart attack
- Death
The TAVR Procedure
What is involved in a TAVR/TAVI procedure?
A TAVR procedure can be executed via small openings that leave all the chest bones intact, contrary to an open heart surgery where the chest is surgically detached for replacing the diseased valve.
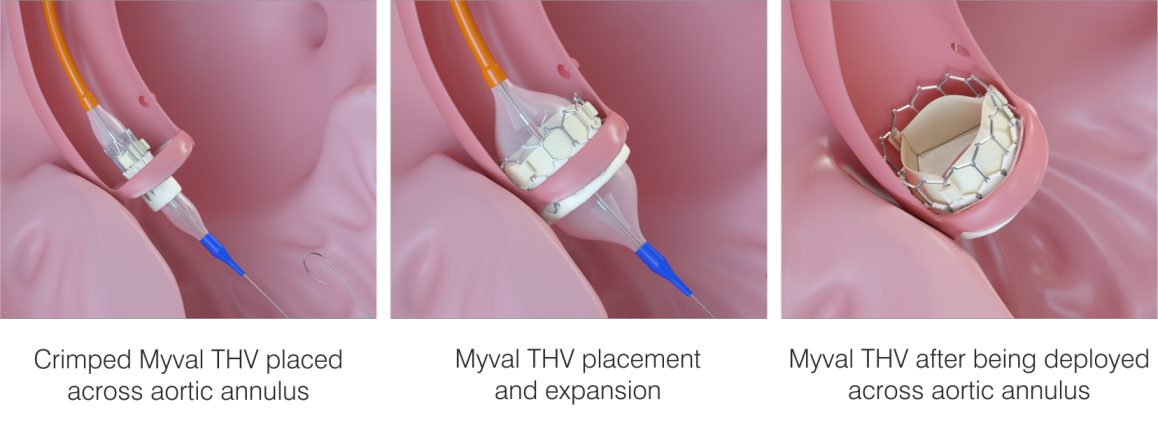
The valves for Transcatheter aortic valve replacement are specifically designed for implantation using a long, narrow tube known as a catheter. The catheter can be inserted through one of the routes explained below:
- Through a large artery in the groin, called the transfemoral approach, not requiring a surgical incision in the chest,
or
- Through a large artery in the chest or through the tip of the left ventricle (the apex), which is known as the transapical approach.
The valve is composed of natural tissue derived from the heart of either a cow or a pig. The natural tissue is re-treated and attached to a flexible expanding mesh frame. The balloon expandable transcatheter heart valve is shown in the figure given below:
How to prepare for TAVR?
The heart team will assess the patient’s general condition through various imaging and blood tests before TAVR surgery, to confirm the condition of the patient’s lungs and heart. The team will also advise a heart ultrasound to the patient to estimate his/her aortic valve. The team may examine his/her heart by performing an angiography.
The patient needs to:
- Inform the doctor about any medicines he/she is taking, including over-the-counter medicines.
- If the patient is habitual to smoking, he/she should question the doctor on how soon before surgery he/she needs to quit.
- If the doctor instructs, you should stop taking certain medicines.
- Stop eating and drinking, generally at 8-12 hours before surgery. This is required as the patient will receive medicines that will make him/her fall asleep/unconscious during the surgery.
What happens during TAVR / TAVI?
Normally, a TAVR procedure follows the following path:
- The patient will be required to do away with any ornaments or other valuables that may delay the procedure or affect its outcomes.
- The patient will also have to remove his/her clothes and has to wear the sterile gowns provided by the hospital staff.
- Patients will have to urinate and/or pass stools before the procedure begins.
- An intravenous (IV) line will be initiated through the patient’s arm or hand.
- Supplementary tubes (catheters) will be inserted in the patient’s neck and wrist to observe the heart’s status and blood pressure or to withdraw blood samples for analysis. Different sites for these catheters include regions beneath the collarbone or the groin.
- The patient will be asked to lie down on the operating table, in supine position on their backs.
- The doctor who gives medication to the patient to block any painful sensation during surgery (anesthesiologist) will monitor the functioning and vital parameters like heartbeat and blood oxygen level nonstop during the surgery.
- Once the patient falls asleep, a breathing tube will be inserted into the patient’s throat and into the windpipe to provide oxygen to his/her lungs.
- During the surgery, the patient will be connected to a machine that will perform the breathing function for the patient (ventilator).
- A catheter will also be introduced into his/her bladder to drain urine during the surgical procedure.
- A transesophageal echocardiogram (TEE) probe will be inserted into his/her food pipe to aid in visualization of the valve.
- The skin of the surgical site in his/her groin will be rinsed with a germ-killing solution.
- The surgeon will initially make a cut in the patient’s groin area to detect the artery carrying blood from the heart down into the leg (femoral artery).
- The surgeon will place a tube/catheter into that artery and reach it to his/her heart and through the aorta, it will reach the patient’s aortic valve.
- The surgeon will also introduce additional catheters in the heart to perform measurements and take X-ray images throughout the procedure.
- The surgeon will guide the artificial valve encased within an expandable balloon apparatus, in to the femoral artery catheter and through the patient’s native diseased aortic valve.
- As soon as the balloon is positioned correctly, the surgeon will enlarge it and implant the artificial valve to replace the diseased valve.
- The surgeon will perform measurements and take images to ensure the new valve is functioning properly, before getting rid of the catheters.
- The doctor will lastly close the artery and the cut in the groin area with sutures.
Recommendations post TAVR procedure and recovery
After the replacement procedure, the patient will be transferred to the intensive care unit (ICU), so as to closely monitor the patient as he/she recovers from surgery. During his/her recovery, the tubes in his/her blood vessels providing nutrition and observing his/her functioning of the heart will be detached. He/she will rapidly gain the capacity to get up and walk and resume taking normal diet on his/her own orally. Even then, the patient may be required to stay in the hospital for some days.
Once the doctors are of the opinion that the patient has recovered adequately to return home, he/she will be cleared from the hospital. Even then, he/she has to follow all the directions for medicines, pain control, diet, activity, and wound care. The patient should also ensure to hold onto all his/her follow-up appointments. Total recovery may take quite a few weeks. Here are some useful guidelines to follow as the patient heals:
- The doctor may prescribe blood thinning medicine to the patient to prevent blood clots after a TAVR/ TAVI. To ensure he/she is receiving the correct dose, he/she may be required to go for regular blood checkups.
- Walk around as much as possible.
- Gradually resume routine activities, but lifting of heavy objects must be avoided.
- Take advice from the doctors on when you can resume driving, work, and sexual activity.
- Observe the groin for any indication of swelling, redness, bleeding, or discharge.
- Inform the doctor if the patient feels any pain or gets a high temperature, bleeding, or difficulty in breathing.
- Consume a heart-healthy diet and maintain a healthy weight.
- Stop smoking.
